Liquid biopsies – an unbiased diagnosis
Scientists in the US have carried out liquid biopsy (as opposed to tissue biopsy) research throughout the summer of 2020 in order to learn how to diagnose cancer through the use of blood tests.
- The tests are referred to as ‘liquid biopsies’. Liquid biopsy is able to detect genetic material from cancer tumours in the bloodstream. Tissue biopsies are localised or limited to one site or sample.
- Liquid biopsies allow for less invasive diagnostic procedures, which ordinarily involve extracting samples from tumours or removing tumours.
- Liquid biopsy tests are also helpful when tumours are located in internal organs and so are difficult or risky to access via surgery.
- Current blood tests are only used for individuals that have already been diagnosed with cancer.
- Blood tests are extremely important as they can tell us all sorts of things about our bodies from cholesterol to infection levels.
- Biopsies, from blood or tissues, are important in determining genetic mutations that cause cancer tumours to grow.
- Liquid biopsy is used in tandem with drugs. This new approach to biopsy also helps with drug choice and ‘design’. The aim is to find choose or design drugs that specifically block certain cancer mutation DNA segments or biochemical pathways.
- So liquid biopsies become important as ‘targeted therapies’ have a tendency to improve survival rates for patients over ‘hammer-to-squash-a-pea’ approaches
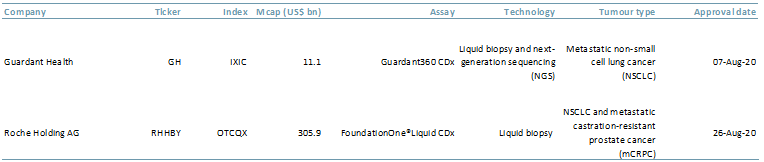
Exhibit 1 illustrates the liquid biopsy tests that were approved in the summer of 2020 by the US Food and Drug Administration along with their technology type and tumours diagnosed.
Exhibit 1 – US FDA liquid biopsy approvals, 2020
 Sources: ACF Equity Research; FDA; Roche
Sources: ACF Equity Research; FDA; Roche
- The Guardant Health (GH:Nasdaq) 360CDx test can track 55 genetic mutations. The GH test can also be used as a ‘companion diagnostic’ in that it can direct doctors to a specific drug for treatment; e.g. AstraZeneca’s osimertinib is specifically used for lung cancer.
- The FoundationOne LiquidCDx from Roche (ROG.SW and RHHBY:PNK) test analyses 300+ cancer-related genes.
- Edward Garon, associate professor of medicine and oncologist at the University of California Los Angeles (UCLA), says that blood biopsies (as opposed to tissue biopsies) help him to keep a closer eye on how his patients are doing, i.e. how the tumours might be changing or if they are developing new mutations that resist medications currently prescribed.
- Liquid biopsies can also provide results a lot faster.
- Ideally, the aim is to screen for cancer much earlier, before it is diagnosed, i.e. getting ahead of the cancer whilst it is relatively easy or before it becomes too advanced to treat.
Research is still ongoing as to the effectiveness of liquid biopsies and when they might be useful or sufficient for a diagnosis.
A University of Texas MD Anderson Cancer Centre 2019 study was carried out on 282 patients with metastatic non-small-cell lung cancer (NSCLC). The metastatic lunger cancer patients provided both tissue and blood biopsies.
The Guardant Health test detected tumour mutations in 77 patients while tissue biopsies detected mutations in only 60 (25% vs. 21%). Liquid biopsy results were provided in nine days compared to the average 15 days for a tissue biopsy.
While the data is encouraging it is still incomplete and insufficient to determine if a liquid biopsy is better than a tissue biopsy.
As a result, continued R&D in healthcare is extremely important if the industry hopes to improve the EMA health-related quality of life (HRQL or QOL in the US) score of patients. However, healthcare companies need resources for R&D, such as technology and funding.
Healthcare R&D spend in 2020 is expected to be USD 185bn. ACF forecasts healthcare R&D to reach $233bn in 2025E, a CAGR of 5%.
These figures present an attractive investment opportunity for investors and a potential for small and mid-cap companies to get a piece of the pie. By way of example, the global liquid biopsy market size is expected to increase to $4.4bn in 2025 up from $0.7bn in 2016, a 515% increase (BIS Research).
In addition to a lucrative investment opportunity, liquid biopsies would have several beneficial short-term market drivers such as higher patient survival rates and lower state/government healthcare costs.
There is also the reduction in cost to insurers (making access to insurance more affordable for everyone else, ceteris paribus) and the value generating extended productivity of individuals, who might otherwise leave the working economy and deprive it of their knowledge and expertise.
A cancer patient’s end-of-life phase is defined as the 12 months before expected death (based on studies of other cancer patients). The end-of-life definition is a cold statistic but end-of-life costs are higher than a patient’s initial care costs and this is even more true of the final 6 weeks.
According to the American Association for Cancer Research (AACR) the end-of-life cost is approximately $105,500 per year per patient.
There is another, perhaps unintended but nevertheless serendipitous, advantage of potential liquid biopsy – it could remove the unconscious ‘ethnic dermatology’ type bias that doctors may have for patients and the symptoms that they exhibit.
In our 29 July 20 blog (Doctors may be missing illnesses because UK medical textbooks often focus on white people) covered the concerns around ‘Ethnic Dermatology’ where patients with pigmented skin tones have a difficult time getting a diagnosis for skin disorders.
It seems the principles described in our ethnic dermatology blog are relevant to other conditions.
Liquid biopsies could provide a potentially more accurate diagnosis regardless of an individual’s ethnicity, sex, or age. This is because we expect a different set of experts to be analysing them compared with tissue biopsy. It is also likely that blood biopsy will be more machine and AI than human.
In addition, a patient could be comforted knowing that the risk their symptoms are dismissed or misinterpreted solely based on their appearance is diminished by a more arms-length or fully automated approach. This comfort also surely speaks to the EMA’s HRQL and the FDA’s quality-of-life tests.
Author: Renas Sidahmed – Renas is a Staff Analyst and part of the Sales & Strategy team at ACF Equity Research. See Renas’s profile here













